Principal Investigator:
M. Flint Beal (deceased)
Background & Unmet Need
- Huntington’s disease (HD) is a dominantly inherited progressive neurodegenerative disease characterized by progressive motor impairment, personality changes, and gradual intellectual decline
- While therapies exist to help treat symptoms, there are currently no approved therapies that stop or reverse decline
- Previous studies have identified a potential link between impairment of the peroxisome proliferator-activated receptor (PPAR)-γ-coactivator 1α (PGC-1 α) levels and activity are HD pathogenesis
- Unmet Need: Disease-modifying therapeutics for the treatment of HD and other neurodegenerative diseases
Technology Overview
- The Technology: Administration of bezafibrate or combinations of PPAR agonists for the treatment of HD and tauopathies
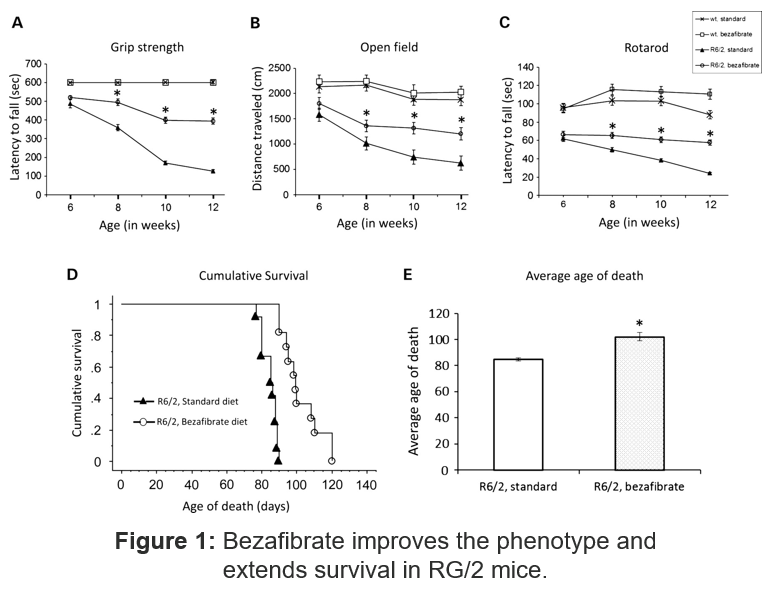
- The Discovery: Bezafibrate improved behavioral impairments, neuronal loss, and prolonged survival in mouse models of HD
- Administration of bezafibrate increased numbers of mitochondria in both brain and muscle tissue
- Bezafibrate treatment also improved the behavioral impairments and tau aggregation in mouse models of tauopathy
Technology Applications
- Repurposing of bezafibrate, or novel combinations of selective PPAR agonists
- Treatment of neurological conditions
Technology Advantages
- Bezafibrate is already approved drug for other indications, which may streamline development
- Potentially applicable to multiple indications
Publications
Resources
Intellectual Property
Patents
- US Patent: 9,592,212. "Use of pan-PPAR agonists for treatment of tauopathies." Issued Mar 14, 2017.
Cornell Reference
- 5283
Contact Information

For additional information please contact
Donna Rounds
Associate Director, Business Development and Licensing
Phone: (646) 780-8775
Email: djr296@cornell.edu

