Principal Investigator:
Ronald G. Crystal, Professor and Chair of Genetic Medicine
Background & Unmet Need
- Allergen-specific Immunoglobulin E (IgE) is integral to the pathogenesis of allergic disorders
- Binding of allergens to IgE antibodies on the surface of mast cells and basophils in sufficient quantities can lead to activation of the allergic response
- IgE-mediated allergic disorders include allergic asthma, allergic rhinitis, atopic dermatitis, and food allergies
- Omalizumab (Xolair) is the only approved anti-IgE therapy, indicated for allergic asthma with ongoing clinical development for peanut allergy
- Omalizumab has numerous limitations, including the need for repeated injections (every 2–4 weeks) and high cost
- Unmet Need: Novel therapies for the management of allergic disorders
Technology Overview
- The Technology: AAV gene therapy that provides sustained delivery of anti-IgE antibodies for the prevention of IgE-mediated allergic reactions
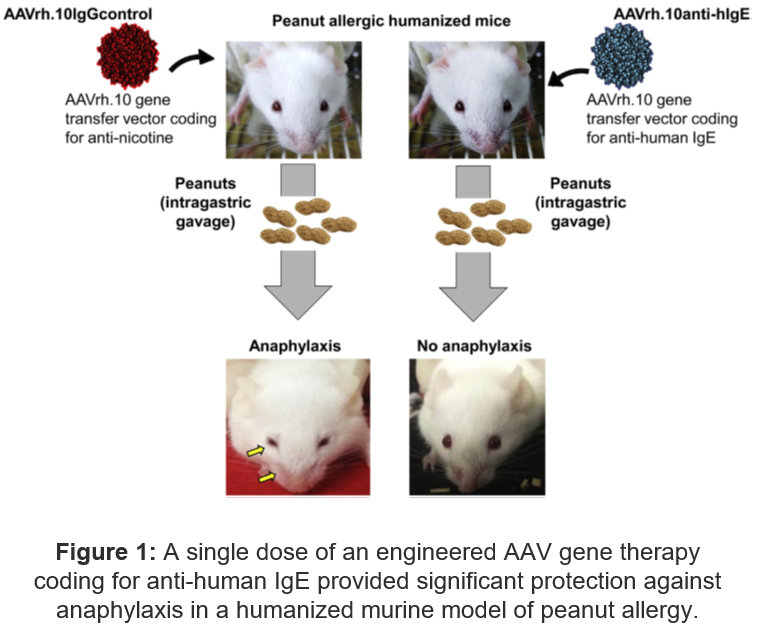
- The DNA sequence encoding for omalizumab was inserted into an AAV gene transfer vector (AAVrh.10anti-hIgE) and introduced into mice
- A humanized murine model of peanut allergy was generated by reconstituting immunodeficient mice with peanut-allergic human blood mononuclear cells
- PoC Data: A single dose of the gene therapy was sufficient for persistent prevention of peanut-induced sever allergy, both for prophylaxis and therapy after mice exhibit the peanut-induced anaphylaxis-related symptomology
- The inventors have also developed novel human anti-IgE antibodies with demonstrated efficacy that are ready to be incorporated into the AAV platform for further testing
Technology Applications
- One-time preventative therapy for peanut allergy and other severe, IgE-mediated allergic disorders
- Gene transfer method could be applied to other agents that block allergic reactions (e.g., soluble IgE receptors, eosinophils, and basophils)
Technology Advantages
- One-time therapy avoids the need for repeated and costly injections of omalizumab
- Durable protection improves patient quality of life by alleviating anxiety and risk associated with travel and dining at restaurants
Publications
Resources
Intellectual Property
Patents
- US Patent 10,293,059. "Gene therapy to prevent reactions to allergens." Issued May 21, 2019.
- JP Patent JP6878301. "Gene therapy to prevent reactions to allergens." Issued May 26, 2021
- CN Patent CN107635584B. "Gene therapy to prevent response to allergens." Issued June 17, 2022.
- CA Patent CA2982213C. "Gene therapy to prevent reactions to allergens." Issued October 18, 2022.
- EP Patent 3,280,445. "Gene therapy to prevent reactions to allergens"
Cornell Reference
- 6985
Contact Information

For additional information please contact
Brian Kelly
Director, Business Development and Licensing
Phone: (646) 825-2766
Email: bjk44@cornell.edu

